ORIGINAL ARTICLE
Effect of Fusarium and Rhizoctonia isolates on the pre-attachment stage of Phelipanche aegyptiaca
1
Plant Pathology, Bati Akdeniz Agricultural Research Institute, Boztepe, 07110, Antalya, Turkey
2
Plant Pathology, Akdeniz University, Pınarbaşı, 07070, Antalya, Turkey
A - Research concept and design; B - Collection and/or assembly of data; C - Data analysis and interpretation; D - Writing the article; E - Critical revision of the article; F - Final approval of article
Submission date: 2025-02-04
Acceptance date: 2025-03-06
Online publication date: 2026-03-20
Corresponding author
Gurkan Basbagci
Plant Pathology, Bati Akdeniz Agricultural Research Institute, Boztepe, 07110, Antalya, Turkey
Plant Pathology, Bati Akdeniz Agricultural Research Institute, Boztepe, 07110, Antalya, Turkey
Journal of Plant Protection Research 2026;1(66):25-36
HIGHLIGHTS
- Biocontrol
- Egyptian broomrape
- parasitic weed
- pre-parasitic stage
KEYWORDS
TOPICS
ABSTRACT
Phelipanche aegyptiaca is an obligate root-parasitic plant of the Orobanchaceae family
that comprises the most threatening weed for major crops in regions with a Mediterranean
climate zone, as well as in Asia, southern and Eastern Europe. The pre-
-attachment stage is crucial for its successful attachment and parasitism, making this
phase a key target for biological control strategies. In this study, the effect of 79 fungal
isolates, including 49 Fusarium and 30 Rhizoctonia, on the pre-attachment stages of
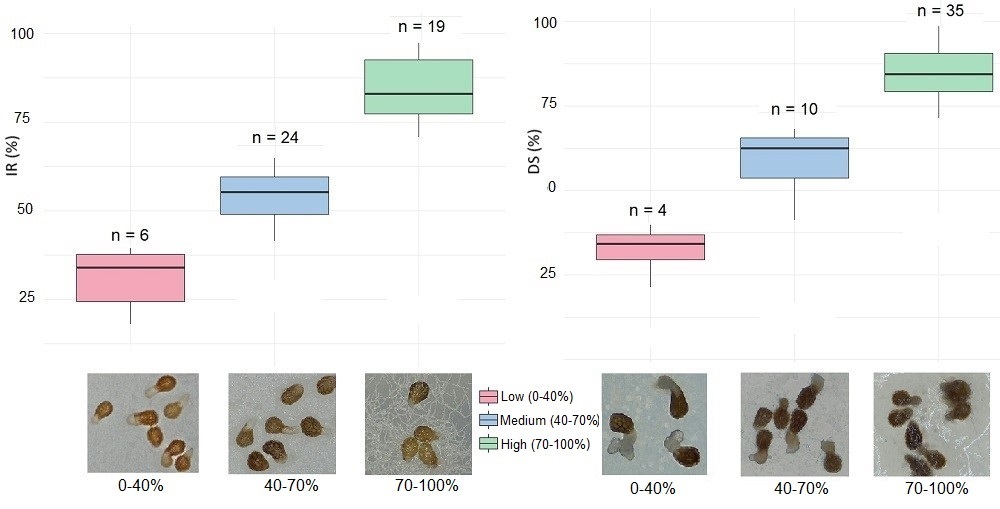
P. aegyptiaca were investigated. According to the experimental results, the Fusarium isolates
inhibited the seed germination by the rate of 17.9 to 97.1%, while the disease severity
values were between 21.3 and 99.8%. Approximately 40% of the 49 Fusarium isolates
exhibited high inhibition effects (70−100%) and around 70% of the isolates caused high
disease severity (70−100%) on the radicle. A positive correlation was observed between
the inhibition rate and disease severity for the Fusarium isolates. Moreover, 15 Fusarium
isolates displayed high effects in both parameters simultaneously. In contrast, the Rhizoctonia
isolates exhibited seed inhibition rates ranging from 1.2 to 37.6% and disease severity
values between 0 and 66.0%, with none showing high effects on any pre-attachment stages.
These findings suggest that the Fusarium isolates have a great potential to use biocontrol
agents against P. aegyptiaca, while the Rhizoctonia isolates have less, which may be due
to differences in their secondary metabolite profiles or other pathogenicity-related traits.
These findings pave the way for future research to optimize Fusarium-based biocontrol
strategies and better understand the broader potential of these fungal isolates in managing
P. aegyptiaca.
FUNDING
This study was supported by the Scientific and Technological
Research Council of Türkiye (TUBITAK)
under Grant Number 123O933. The authors thank
TUBITAK for their support.
RESPONSIBLE EDITOR
Iwona Adamska
CONFLICT OF INTEREST
The authors have declared that no conflict of interests exist.
REFERENCES (59)
1.
Abbas H.K., Boyette C.D., Hoagland R.E. 1995. Phytotoxicity of Fusarium, other fungal isolates, and of the phytotoxins fumonisin, fusaric acid, and moniliformin to jimsonweed. Phytoprotection 76 (1): 17–25. DOI: https://doi.org/10.7202/706081....
2.
Abbott W.S. 1925. A method of computing the effectiveness of an insecticide. Journal of Economic Entomology 18 (2): 265–267. DOI: https://doi.org/10.1093/jee/18....
3.
Abouzeid M.A., El-Tarabily K.A. 2010. Fusarium spp. suppress germination and parasitic establishment of bean and hemp broomrapes. Phytopathologia Mediterranea 49 (1): 51–64. DOI: https://doi.org/10.14601/Phyto....
4.
Adams G.C. 1996. Genetics of Rhizoctonia species. p. 101–116. In: “Rhizoctonia species: Taxonomy, Molecular Biology, Ecology, Pathology and Disease Control” (B. Sneh, S. Jabaji-Hare, S. Neate, G. Dijst, eds). Springer, 594 pp.
5.
Ajayi‐Oyetunde O., Bradley C. 2018. Rhizoctonia solani: taxonomy, population biology and management of rhizoctonia seedling disease of soybean. Plant Pathology 67 (1): 3–17. DOI: https://doi.org/10.1111/ppa.12....
6.
Aksoy E., Uygur F. 2008. Effect of broomrapes on tomato and faba bean crops. Türkiye Herboloji Dergisi 11 (1): 1–7.
7.
Andersen T.F., Rasmussen H.N. 1996. The mycorrhizal species of Rhizoctonia. p. 379–390. In: “Rhizoctonia species: Taxonomy, Molecular Biology, Ecology, Pathology and Disease Control” (B. Sneh, S. Jabaji-Hare, S. Neate, G. Dijst, eds). Springer, 594 pp.
8.
Bacon C., Porter J., Norred W., Leslie J. 1996. Production of fusaric acid by Fusarium species. Applied and Environmental Microbiology 62 (11): 4039–4043. DOI: 10.1128/aem.62.11.4039-4043.1996.
9.
Bai J.R., Wei Q., Shu J.S. Gan Z.X., Li B.J., Yan D.L., Huang Z.J., Guo Y.M., Wang X.X., Zhang L.X., Cui Y.N., Lu X.X., Lu J.H., Pan C.Y., Hu J.L., Du Y.C., Liu L., Li J.M. 2020. Exploration of resistance to Phelipanche aegyptiaca in tomato. Pest Management Science 76 (11): 3806–3821. DOI: https://doi.org/10.1002/ps.593....
10.
Bailey B.A. Apel-Birkhold P.C., Akingbe O.O., Ryan J.L., O'Neill N.R., Anderson J.D. 2000. Nep1 protein from Fusarium oxysporum enhances biological control of opium poppy by Pleospora papaveracea. Phytopathology 90 (8): 812–818. DOI: https://doi.org/10.1094/PHYTO.....
11.
Başbağci G., Unal F., Uysal A., Dolar F.S. 2019. Identification and pathogenicity of Rhizoctonia solani AG–4 causing root rot on chickpea in Turkey. Spanish Journal of Agricultural Research 17 (2): 1007. DOI: 10.5424/sjar/2019172-13789.
12.
Başbağci G., Cignitas E., Kitis Y.E. 2023. Fungi isolated from broomrape species (Phelipanche spp.) in tomato greenhouses of the Western Mediterranean Region. Turkish Journal of Weed Science 26 (2): 106–113 (in Turkish).
13.
Başbağcı G., Çiğnitaş E., Kitiş Y.E. 2024. Diversity and geographic distribution of fungi on broomrape species. Turkish Journal of Weed Science 27 (1): 49–75.
14.
Bendejacq‐Seychelles A., Gibot‐Leclerc S., Guillemin J.P., Mouille G., Steinberg C. 2024. Phytotoxic fungal secondary metabolites as herbicides. Pest Management Science 80 (1): 92–102. DOI: https://doi.org/10.1002/ps.781....
15.
Bhuiyan M., Mendoza L.D.R., Lakshman D.K., Qi A., Khan M. 2024. Effects of inoculum density of R. solani AG 2–2IIIB and age of plant on root rot severity in sugar beet. Journal of Plant Protection Research 65 (1): 125–132. DOI: 10.24425/jppr.2025.153824.
16.
Boari A., Vurro M. 2004. Evaluation of Fusarium spp. and other fungi as biological control agents of broomrape (Orobanche ramosa). Biological Control 30 (2): 212–219. DOI: https://doi.org/doi:10.1016/j.....
17.
Bouwmeester H.J., Matusova R., Zhongkui S., Beale M.H. 2003. Secondary metabolite signalling in host-parasitic plant interactions. Current Opinion in Plant Biology 6 (4): 358–364. DOI: https://doi.org/https://doi.or....
18.
Boyette C.D., Quimby Jr P.C., Connick Jr W.J., Daigle D.J., Fulgham F.E. 1991. Progress in the production, formulation, and application of mycoherbicides. p. 209–222. In: “Microbial Control of Weeds” (TeBees D.O., ed.). Springer New York, 284 pp. DOI: https://doi.org/10.1007/978-1-....
19.
Boyette C.D., Quimby Jr P., Caesar A., Birdsall J., Connick Jr W., Daigle D., Jackson M., Egley G., Abbas H. 1996. Adjuvants, formulations, and spraying systems for improvement of mycoherbicides. Weed Technology 10 (3): 637–644. DOI: 10.1017/S0890037X00040562.
20.
Breslow N. 1970. A generalized Kruskal-Wallis test for comparing K samples subject to unequal patterns of censorship. Biometrika 57 (3): 579–594. DOI: https://doi.org/10.2307/233477....
21.
Brun G., Braem L., Thoiron S., Gevaert K., Goormachtig S., Delavault P. 2018. Seed germination in parasitic plants: what insights can we expect from strigolactone research? Journal of Experimental Botany 69 (9): 2265–2280. DOI: https://doi.org/10.1093/jxb/er....
22.
Capasso R., Evidente A., Cutignano A., Vurro M., Zonno M.C., Bottalico A. 1996. Fusaric and 9, 10–dehydrofusaric acids and their methyl esters from Fusarium nygamai. Phytochemistry 41 (4): 1035–1039. DOI: https://doi.org/10.1016/0031-9....
23.
Cardoso C., Ruyter-Spira C., Bouwmeester H.J. 2011. Strigolactones and root infestation by plant-parasitic Striga, Orobanche and Phelipanche spp. Plant Science 180 (3): 414–420. DOI: https://doi.org/https://doi.or....
24.
Cignitas E., Basbagci G., Sulu G., Kitis Y.E. 2024. Fusarium fujikuroi as a potential biocontrol agent of the parasitic weed Phelipanche aegyptiaca in tomato. Journal of Phythopathology 172 (3): e13344. DOI: https://doi.org/DOI: 10.1111/jph.13344.
25.
Cignitas E., Kitis Y.E. 2022. Molecular identification of Phelipanche species from the western Mediterranean region of Türkiye. 19th European Weed Research Society Symposium, 20–23 June 2022, Athens, Greece.
26.
Core R. 2015. Team. R: a language and environment for statistical computing, 2021.
27.
Delavault P., Montiel G., Brun G., Pouvreau J.-B., Thoiron S., Simier P. 2017. Communication between host plants and parasitic plants. Advances in Botanical Research 82: 55–82. DOI: https://doi.org/10.1016/bs.abr....
28.
Desjardins A.E., Hohn T.M. 1997. Mycotoxins in plant pathogenesis. Molecular Plant-Microbe Interactions 10 (2): 147–152.
29.
Dor E., Evidente A., Amalfitano C., Agrelli D., Hershenhorn J. 2007. The influence of growth conditions on biomass, toxins and pathogenicity of Fusarium oxysporum f. sp. orthoceras, a potential agent for broomrape biocontrol. Weed Research 47 (4): 345–352. DOI: https://doi.org/10.1111/j.1365....
30.
Dor E., Hershenhorn J. 2009. Evaluation of the pathogenicity of microorganisms isolated from Egyptian broomrape (Orobanche aegyptiaca) in Israel. Weed Biology and Management 9 (3): 200–208. DOI: https://doi.org/10.1111/j.1445....
31.
Dor E., Hershenhorn J., Andolfi A., Cimmino A., Evidente A. 2009. Fusarium verticillioides as a new pathogen of the parasitic weed Orobanche spp. Phytoparasitica 37 (4): 361–370. DOI: https://doi.org/10.1007/s12600....
32.
Duafala T., Wilhelm S., Gold A., Sagen J. 1976. Rhizoctonia disease of broomrape, a possible biological control. The American Phytopathological Society 3: 272.
33.
Eizenberg H., Goldwasser Y. 2018. Control of Egyptian broomrape in processing tomato: A summary of 20 years of research and successful implementation. Plant Disease 102 (8): 1477–1488. DOI: https://doi.org/10.1094/PDIS-0....
34.
FAOSTAT. 2022. Crops and livestock products. Retrieved 10.03.2024 from https://www.fao.org/faostat/en....
35.
Fernández-Aparicio M., Reboud X., Gibot-Leclerc S. 2016. Broomrape weeds. Underground mechanisms of parasitism and associated strategies for their control: a review. Frontiers in Plant Science 7: 135. DOI: https://doi.org/10.3389/fpls.2....
36.
Ghannam I., Barakat R., Al-Masri M. 2007. Biological control of Egyptian broomrape (Orobanche aegyptiaca) using Fusarium spp. Phytopathologia Mediterranea 46 (2): 177–184. DOI: https://doi.org/10.14601/Phyto....
37.
Gibot-Leclerc S., Guinchard L., Edel-Hermann V., Dessaint F., Cartry D., Reibel C., Gautheron N., Bernaud E., Steinberg C. 2022. Screening for potential mycoherbicides within the endophyte community of Phelipanche ramosa parasitizing tobacco. Fems Microbiology Ecology 98 (3). DOI: https://doi.org/10.1093/femsec....
38.
Goldwasser Y., Rodenburg J. 2013. Integrated Agronomic Management of Parasitic Weed Seed Banks. In: “Parasitic Orobanchaceae” (Joel D., Gressel J., Musselman L., eds.). Springer, Berlin, Heidelberg. DOI: https://doi.org/10.1007/978-3-....
39.
Hameed K., Saadoun I., Shyab Z.A. 2001. Potential biological control of Orobanche by fungi isolated from diseased specimens in Jordan. The Plant Pathology Journal 17 (5): 257–263.
40.
Herr L.J. 1995. Biological control of Rhizoctonia solani by binucleate Rhizoctonia spp. and hypovirulent R. solani agents. Crop Protection 14 (3): 179–186. DOI: https://doi.org/10.1016/0261-2....
41.
Joel D., Hershenhorn J., Eizenberg H., Aly R., Ejeta G., Rich P., Ransom J., Sauerborn J., Rubiales D. 2007. Biology and management of weedy root parasites. Horticultural Reviews 33: 267–349. DOI: https://doi.org/10.1002/978047....
42.
Karam Pur F., Fasihi M., Heydari A. 2004. Identification of fungal pathogenic agents of broomrape on tomato fields AGRIS – International System for Agricultural Science and Technology. Available on: https://agris.fao.org/search/e... [Accessed: May, 2024].
43.
Lemańczyk G., Lisiecki K., Piesik D. 2023. Binucleate Rhizoctonia strain: a potential biocontrol agent in wheat production. Agronomy 13 (2): 523. DOI: https://doi.org/10.3390/agrono....
44.
Macías-Rubalcava M.L., Garrido-Santos M.Y. 2022. Phytotoxic compounds from endophytic fungi. Applied Microbiology and Biotechnology 106 (3): 931–950. DOI: https://doi.org/10.1007/s00253....
45.
Maculewicz D. 2015. Binucleate Rhizoctonia spp. as a biocontrol agents against plant pathogens. Ecological Chemistry and Engineering A: 22 (2): 195–203. DOI: 10.2428/ecea.2015.22(2)16.
46.
Misra V.A., Wafula E.K., Wang Y., Depamphilis C.W., Timko M.P. 2019. Genome-wide identification of MST, SUT and SWEET family sugar transporters in root parasitic angiosperms and analysis of their expression during host parasitism. BMC Plant Biology 19: 1–18. DOI: https://doi.org/10.1186/s12870....
47.
Parker C. 2012. Parasitic weeds: a world challenge. Weed Science 60 (2): 269–276. DOI: https://doi.org/10.1614/WS-D-1....
48.
Pitrat M., Risser G., Epinat C., Ferrière C., Ricard M., Olivier C., RuYnato A., Lecoq H., Blancard D., Bertrand F., Nicot A., Glandard A., Molot P., Mas P. 1991. Techniques d’inoculation artificielle du melon avec différents agents pathogènes pour la sélection de variétes résistantes. Informal Technical Bulletin Edited by INRA, Montfavet, Station d’Amélioration des Plantes maraîchères and Station de Pathologie végétale.
49.
Rispail N., Dita M.A., González‐Verdejo C., Pérez‐de‐Luque A., Castillejo M.A., Prats E., Román B., Jorrín J., Rubiales D. 2007. Plant resistance to parasitic plants: molecular approaches to an old foe. New Phytologist 173 (4): 703–712. DOI: https://doi.org/10.1111/j.1469....
50.
Sneh B., Jabaji-Hare S., Neate S., Dijst G. 2013. Rhizoctonia Species: Taxonomy, Molecular Biology, Ecology, Pathology and Disease Control. Springer Science and Business Media, 578 pp.
51.
Taheri P., Hosseini-Zahani F., Tarighi S. 2024. Binucleate Rhizoctonia induced tomato resistance against Rhizoctonia solani via affecting antioxidants and cell wall reinforcement. Heliyon 10 (6). DOI: https://doi.org/10.1016/j.heli....
52.
Thomas H., Sauerborn J., Muller-Stover D., Kroschel J. 1999. Fungi of Orobanche aegyptiaca in Nepal with potential as biological control agents. Biocontrol Science and Technology 9 (3): 379–381. DOI: https://doi.org/10.1080/095831....
53.
Townsend G.R., Heuberger J.W. 1943. Methods for estimating losses caused by diseases in fungicide experiments. Plant Disease Reporter 27: 340–343.
54.
Vurro M., Prandi C., Baroccio F. 2016. Strigolactones: how far is their commercial use for agricultural purposes? Pest Management Science 72 (11): 2026–2034. DOI: https://doi.org/10.1002/ps.425....
55.
Watson A.K. 2013. Biocontrol. p. 469–490. In: “Parasitic Mechanisms and Control Strategies” (Joel D.M., Gressel J., Musselman L.J., eds). Springer Heidelberg New York Dordrecht London, 513 pp. DOI 10.1007/978-3-642-38146-1.
56.
Wickham H. 2009. ggplot2: elegant graphics for data analysis New York. NY: Springer.
57.
Xu D., Xue M., Shen Z., Jia X., Hou X., Lai D., Zhou L. 2021. Phytotoxic secondary metabolites from fungi. Toxins 13 (4): 261. DOI: https://doi.org/10.3390/toxins....
58.
Yoshida S., Cui S., Ichihashi Y., Shirasu K. 2016. The haustorium, a specialized invasive organ in parasitic plants. Annual Review of Plant Biology 67 (1): 643–667.
59.
Zrenner R., Genzel F., Verwaaijen B., Wibberg D., Grosch R. 2020. Necrotrophic lifestyle of Rhizoctonia solani AG3-PT during interaction with its host plant potato as revealed by transcriptome analysis. Scientific Reports 10 (1): 12574. DOI: https://doi.org/10.1038/s41598....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.